New mechanism for identification of future dysfunctional cells can help in spotting autism & early detection of cancer
A group of researchers has developed a mechanism capable of early identification of molecular changes that may lead to dysfunctional cells in the human body. This vital information, the scientists say, can be useful in detection of autoimmune diseases like autism and the early detection of cancer, thereby aid in planning better therapeutic strategies.
Early diagnosis holds key in initiating medical treatments for most diseases. But often, the disease condition is identified only after it has reached advanced stages, thus delaying the process of treatment, recovery, and cure.
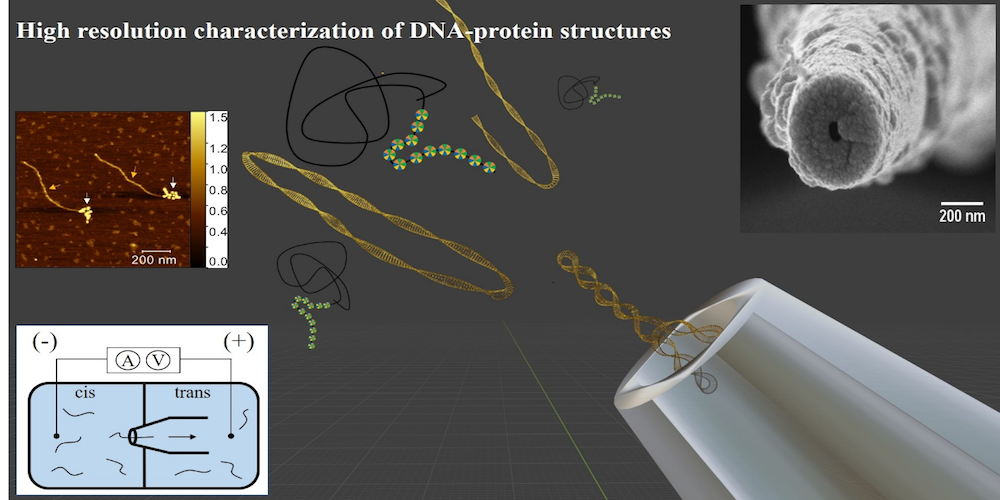
In the present study, published in the journal ACS Sensors of the American Chemical Society, the research team from Raman Research Institute (RRI), an autonomous institution of the Department of Science and Technology (DST), used nanopore devices to decipher the structures of nucleosomes which are responsible to regulate the genetic and epigenetic information within the cell’s nucleus. Nucleosomes are the basic structural unit of DNA packaging. The team led by Professor Gautam Soni, comprising Sumanth Kumar, Divya Shet, Serene David, and Mahesh B.L., recently published their findings in a research paper titled ‘Nanopore sensing of DNA – Histone Complexes on Nucleosome Arrays’.
“Being highly sensitive, technology using nanopore devices can be deployed to detect disease-specific biomarkers on the DNA. They can be detected even if they are present in low concentrations as observed during the early disease onset stage. This could then bridge the gap in the practice of epigenetic diagnostics,” said Prof. Gautam Soni.
The study holds the potential in deepening the present understanding of DNA packaging and cell functioning in the human body. Besides, the experimental technique can be replicated in the form of a portable molecular diagnostic device for disease-specific screening for biomarkers.
For the experiment, scientists dipped a glass capillary with a few nanometre-sized opening (nanopore) into an ionic solution. When subjected to voltage, the ions present in the medium seamlessly passed through the nanopore. The high-resolution measurement of subtle fluctuations in the current when the analyte particles (like DNA and/or proteins) passed through the nanopore allowed the team to detect as well as confirm changes in the molecular size of nucleosomes.
The researchers said that nanopore is reliable and can carry out large-scale screenings of molecule / protein/ DNA interactions, thus providing information about any epigenetic modifications that occur within cells.
Understanding the stability of the nucleosomes with respect to the epigenetic modifications is essential to obtain the full understanding of key forces that help chromatin maintain its structure which in turn indicates the state of the health of the cells.



[…] New mechanism for identification of future dysfunctional cells can help in spotting autism & ear… […]